What is a Conductivity Meter?
A conductivity meter is used to measure electrical current (or) conductance in solutions.
- What is a Conductivity Meter?
- What is SI unit of conductivity?
- What is Symbol of Conductivity?
- What is the Fundamental Concept of Conductivity Meter?
- What is the Scientific Name of Conductivity Meter?
- What is the Purpose of Conductivity Meter?
- Conductivity Meter with Electromagnetic Induction Method
- How to use a Conductivity Meter?
- Advantage of Conductivity Meter
- Disadvantage of Conductivity Meter
- Applications of Conductivity Meter
Conductivity is a key factor in determining the overall condition of a natural water body.
In water treatment plants, it is additionally used to track changes in the way wastewater is handled.

What is SI unit of conductivity?
The SI unit for conductivity is Siemens per meter (S/m).
What is Symbol of Conductivity?
The sign for electrical conductivity:

What is the Fundamental Concept of Conductivity Meter?
Conductivity refers to a material’s ability to convey an electrical current. To assess conductivity instruments use the following principle:
two plates are inserted in the sample
1). A potential (usually a sine wave voltage) is applied across them and
2). A amount of current flowing via the mixture is measured.
k = K/R
In this equation,
k represents the conductivity of the solution,
K is the constant determined by the electrode and
R is the solution resistance.
What is the Scientific Name of Conductivity Meter?
A solution’s electrical conductivity is tested with an electrical conductivity meter.
It is frequently utilized in
- Hydroponic,
- Aquaculture,
- Aquaponic &
- Freshwater systems
to measure the concentration of nutrients, salts and contaminants in the water.
It has several applications in science and engineering.
What is the Purpose of Conductivity Meter?
Conductivity refers to a material’s ability to conduct electricity (as in solutions, metals, or gasses).
Even though all materials may conduct electric currents their strength may vary.
A conductivity meter operates by using a portable probe for field (or) on-site testing.
After inserting the probe into the liquid to be tested the meter transmits voltage between 2 electrodes within it.
The solution’s electrical resistance causes a voltage decrease which the meter detects. The meter converts this reading to milli, micro (or) micro-Siemens per cm.
This quantity refers to all dissolved solids.
Total dissolved solids refers to the number of particles that can pass through a glass fibre filter.
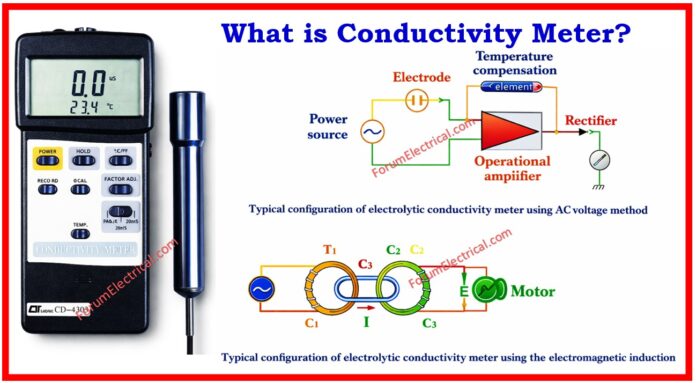
There are two types of conductivity measurement methods:
1). Alternating Bipolar Conductivity Meter
2). Electromagnetic Induction Conductivity Meter.
The Alternating Bipolar Method is used to measure electrolytic conductivity by immersing a two-pole electrode in a solution.
The two poles are connected to an AC power source. The electric current going between the poles is used to quantify both the solution’s resistance (R) and its electrolytic conductivity.
Because electrolytic conductivity varies with temperature, a temperature adjustment device is utilized to match the observed conductivity to that of 25°C.
Conductivity Meter with Electromagnetic Induction Method
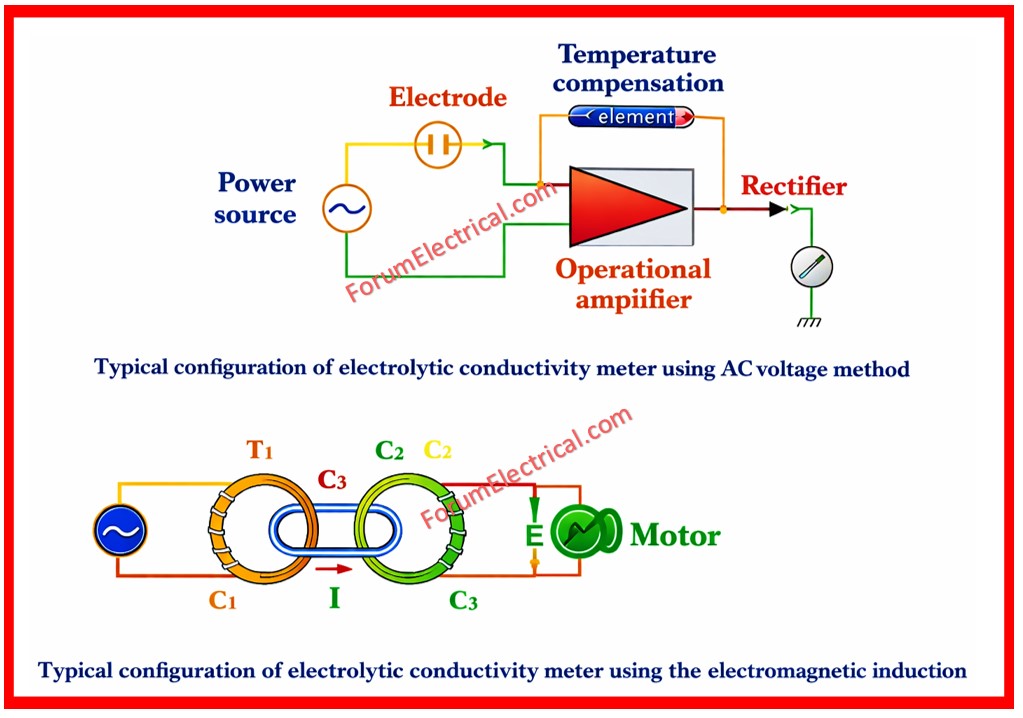
Transformers T1 as well as T2 are utilized to produce two electrodes that are immersed in a solution.
When an alternating current (AC) is delivered to the primary coil C1 an induced current (I) proportional to the electrolytic conductivity of solution passes through C2.
When the transformer T2 employs coil C2 as its primary coil, the secondary coil C3 generates voltage E proportional to the induced current I.
This voltage is used to measure electrolytic conductivity.
How to use a Conductivity Meter?
When using a conductivity meter, clean and connect the probe & calibrate it using standard solutions then submerge it into your sample (gently stir), observe for the measurement to stabilize and record the result in milliSiemens (mS/cm) or microSiemens (µS/cm) and always rinsing with distilled water in between measurements.
Accuracy is impacted by temperature correction and unit observation.
Advantage of Conductivity Meter
1). One advantage of conductivity meters is their portability making them easy to use in labs and workplaces.
2). The conductivity meter processes data using a microprocessor, and its detection accuracy is quite good.
3). The results are easily examined on an LCD screen allowing an inspector to do so with minimal effort.
4). Conductivity meters feature attachments that are simple to connect to a wide range of automation systems making automation feasible.
5). Since the test sample remains intact throughout and prior to the test it is a very cost-effective non-destructive testing technique.
Disadvantage of Conductivity Meter
1). One disadvantage of conductivity meters is their inability to differentiate between ion types.
2). Conductivity meters are temperature-dependent; conductance increases by approximately 2% every degree Celsius.
3). Conductivity meters do not reliably measure ions in a solution.
Applications of Conductivity Meter
1). Conductivity measurement is a versatile tool for controlling processes.
2). An EC meter is used to assess the homogeneity of alloy components.
3). An electrical conductivity meter may also be utilized to calculate a material’s current resistance.
4). An EC meter may be utilized to test the conductivity of nonferrous metals such as copper and aluminum.
5). An electrical conductivity meter monitors the heat treatment process during metal hardening and heat working.